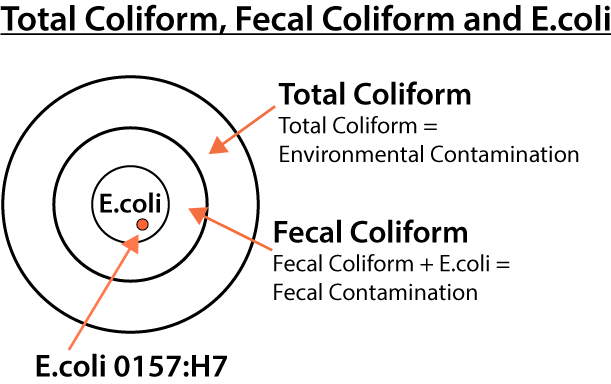
Coliforms have historically been an important group of bacteria in food microbiology, for their practical role in serving as indicator organisms for possible faecal contamination and even E. coli contamination. This piece of writing was completed by Suryani Tan, one of my 2nd-year food microbiology students at The University of Melbourne in 2007.

In order to be valuable as an assessor of faecal contamination, indicator organisms should satisfy the following criteria (Chapman and Hall 1997):
• They should be a member of the normal intestinal flora of healthy people.
• They should be exclusively intestinal in habitat and therefore exclusively faecal in origin if found outside the intestine.
• Ideally they should only be found in humans.
• They should be present when faecal pathogens are present and only when faecal pathogens are expected to be present.
• They should be present in greater numbers than the pathogen they are intended to indicate.
• They should be unable to grow outside the intestine with a die-off rate slightly less than the pathogenic organism.
• They should be resistant to natural environmental conditions and to water and wastewater treatment processes in a manner equal to or greater than the pathogens of interest.
• They should be easy to isolate, identify and enumerate.
• They should be non-pathogenic
The finding of an indicator organism in a properly treated water indicates the presence of material of faecal origin and thus potential contamination (Chapman and Hall 1997).
All of the above requirements are fulfilled by the coliform group. Coliform has been the principle means by which the sanitary quality of water is determined. Several other organism such as faecal Streptococci and Clostridium perfringens are also used to provide supplementary information in certain circumstances as they also meet many of these requirements (Chapman and Hall 1997).
Development of Coliforms as indicator organisms
Direct isolation of specific pathogenic organisms were used as the earliest methods of water bacteriology. However, some difficulties were encountered with the detection of typhoid-like organisms. Bacteriologists attempting to overcome the difficulty of isolating such organisms found that human faeces contained large numbers of aerobic, Gram-negative organisms that resemble typhoid bacteria (Chapman and Hall 1997).
These bacteria were subsequently found to be bile salt-tolerant, facultatively anaerobic organisms capable of growth and fermenting lactose at 37 degree Celsius. These bacteria are known as colifoms. Coliforms were quickly recognised as possible indicators of faecal pollution and procedures were developed to detect and estimate their numbers in raw waters. The most widely adopted method of isolation was inoculation of MacConkey Broth and incubation at 37 degree Celsius for 48 hours. Cultures producing acid and gas were subsequently subcultured onto MacConkey Agar to eliminate obligate anaerobes. A coliform became any organism that was isolated by this method. Faecal colifoms could produce gas from glucose at 47 degree Celsius while coliforms from non-faecal sources failed to grow at these temperatures. E.coli was also capable of growth and fermenting lactose at this temperature with the production of gas and producing indole from tryptophan (Chapman and Hall 1997).
Definition of the coliform group
The coliform group consists of several genera of bacteria belongings to the family Enterobacteriaceae. These genera included Escherichia, Citrobacter, Enterobacter and Klebsiella. The definition of the coliform group has been based on methods used for its detection rather than on the tenets of systematic bacteriology (Chapman and Hall 1997).
Sources of coliforms in milk and how pathogenic they are
Gram-negative bacteria are the agents most often isolated from acute clinical cases of mastitis. The term coliform mastitis is incorrectly used to identify mammary disease caused by all Gram-negative bacteria. The Genera classified as coliforms are Escherichia, Klebsiella, and Enterobacter.Gram-negative bacteria are considered environmental mastitis pathogens. One of the sources of coliforms contaminants in milk is through the transfer of Gram-negative bacteria from the mammary glands of infected cows to uninfected cows (Hogan and Smith 2003). However, this process appears minimal when compared with the constant environmental exposure. Coliform bacteria occupy many habitats in the cow’s environment. Escherichia coli are the normal inhabitants of the gastrointestinal tract of warm-blooded animals. Both Klebsiella and Enterobacter species populate soils, grains, water, and intestinal tracts of animals. Gram-negative bacteria are usually isolated from any surface of the cow or her surroundings and cause other diseases (Hogan and Smith 2003). Coliform bacteria are among the aetiological agents commonly responsible for infectious respiratory and urogenital diseases in dairy cows. Although the mammary gland is not considered a natural habitat for coliform bacteria, many strains are capable of surviving and multiplying in the mammary gland (Hogan and Smith 2003).
Endotoxin
Endotoxin is the primary virulence factor of Gram-negative bacteria responsible for damage to the cow. Endotoxin refers to the lipopolysaccharide portion of the Gram-negative bacterial wall. Endotoxin is released from the bacteria at the time of cell death initiating an inflammatory response. Endotoxin does not directly affect secretory cell but distrupts the blood flow (Shuster 1991 cited in Hogan and Smith 2003). Signs of clinical mastitis include anorexic, fever, dehydration and diarrhea. Decreased milk production during clinical coliform mastitis results both directly and indirectly from the local and systematic effects of endotoxin. Coliform mastitis can result is bacteremia and septicemia as the blood-milk barrier is destroyed. Septicemia is rare but often fatal when it occurs (Hogan and Smith 2003).
Gram-negative bacteria are the leading cause of clinical mastitis in well-managed dairies with low bulk tank milk somatic cell counts. Coliform pathogens account for the majority of peracute cases of clinical mastitis in a herd (Hogan and Smith 2003).
Dry Period
Rates of new intramammary infections caused by coliforms are greater during the dry period than during lactation. During the dry period, susceptibility to intramammary infections is greatest the two weeks after drying off and the two weeks prior to calving. Many infections acquired during the dry period persist to lactation and become clinical cases. 65% of coliform clinical cases that occur in the first two months of lactation are intramammary infections that originated during the dry period. E.coli infections present at calving and early lactation most often originate during the last two weeks of the dry period (Hogan and Smith 2003).
Lactation
Coliform intramammary infections’ rate during lactation is highest at calving and decreases as days in milk advances. The average duration of E. coli intramammary infections during lactation is less than ten days. A major difference between intramammary infections caused by coliform bacteria and those caused by other Gram-negative bacteria is the duration that bacteria persist in the mammary gland. Intramammary infections caused by Serratia and Pseudomonas species often are chronic infections that may persist multiple lactations. Intramammary infections caused by Gram-negative bacteria seldom exceeds 5% of quarters in a herd, however greater than 25% of cows in well-managed herds are diagnosed yearly with clinical mastitis caused by coliforms. Therefore, these diseases caused by these bacteria is seldom great enough to cause bulk tank somatic cell counts (SCC) greater than 400 000/ml but approximately 85% of coliform infections will cause clinical mastitis. Hence, even low SCC herds can still have mastitis problems. The rate of clinical cases caused by Gram-negative bacteria average about 20 cases per 100 cows in a year. The severity of clinical cases caused by coliform bacteria ranges from mild local signs to severe systemic involvement. The majority of clinical coliform cases are characterized by abnormal milk and swollen gland. Only about 10% of clinical coliform cases results in systemic signs including fever, anorexic, and altered respiration. Furthermore, coliform bacteria can also cause peracute mastitis (Hogan and Smith 2003).
References
Chapman and Hall. 1997. Coliform index and water-borne diseases. TJ Press: Great Britain.
Hogan, J and Smith, K, L. 2003. Coliform mastitis. Vetenary Research 34:507-519.
Shuster D.E., Harmon R.J., Jackson J.A., Hemken R.E., Suppression of milk production during endotoxin-induced mastitis, J. Dairy Sci. 74 (1991) 3746–3774.