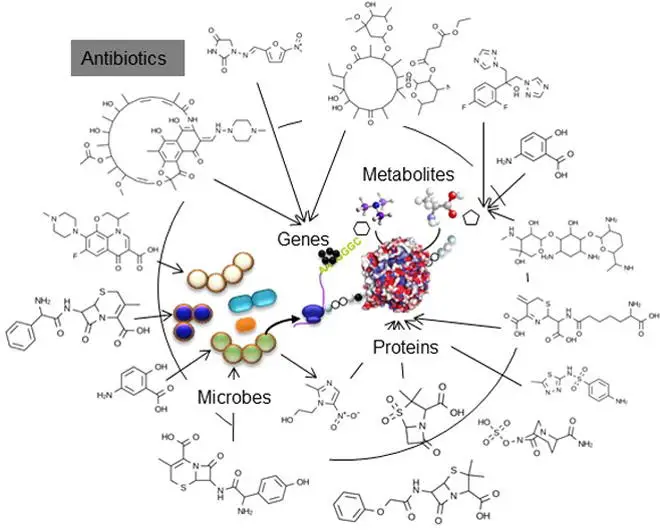
Antibiotics, discovered and commercially developed in the 1930s, have gone from being hailed as ‘magic bullets’ in 1940s human medical practice to nearly a scourge of humanity today, through decades of overuse and misuse in human and veterinary medicine. As the World Health Organization states, they really do pose a big public health threat, across the world (Figure 1). Despite being the foundation of ‘miracle’ cures in the early days, those gains in the war against human bacterial diseases are starting to unravel and are at serious risk of being reversed. Not only that, but residual levels of antibiotics present in the food supply, resulting from widespread use in the agricultural environment has posed additional threats to human health. These represent another facet of food safety, apart from the more common microbial food safety, which has wide-ranging public health consequences. Seemingly, the lack of understanding around the molecular genetics of antibiotic resistance development and consumers’ comfort level with antibiotics, means that the general public appears to have very little concern for the environmental, personal health and public health consequences of antibiotic overuse and misuse. Interestingly, the same people are quite possibly staunchly opposed to genetic modification in agriculture, which has no safety concerns, yet are comfortable with antibiotics, despite their negative impacts on the environment and on humanity. In this article, I want to focus on the effects of antibiotics on the gut microbiome and the subsequent negative on that may arise for one’s personal health, which may or may not be able to be rectified through consumption of probiotic foods. A further consequence could be increased susceptibility to foodborne disease.

One’s gut microbiome, which develops shortly after birth, is a delicately balanced ecosystem of specific bacterial species characteristic of that individual. While some generalisations can be made, the finer aspects are unique to each of us. Overall, we still have much to learn and understand about the human gut microbiome (Iizumi, 2017). However, what we are increasingly gaining an acknowledgement of, and indeed an appreciation for, are the diverse impacts good gut health may have for it’s host. Conversely, the negative consequences of disruption to our specific balance of bacterial species can have serious detriment.
While antibiotics are indeed ‘wonder drugs’ capable of eliminating or stopping the growth of many bacteria, there are downsides. I’m not just talking about contribution to the devastation of antibiotic resistance prevailing in the world, or the side effects to antibiotic therapy an individual can experience, but to the disruption or sometimes decimation to one’s gut microbiome that may result during a course of antibiotics. These are potentially big problems of a personal nature for us.
It is acknowledged that the gut microbiome, and in fact our natural flora in other parts of the body, plays diverse positive roles in our personal health. Originally this was thought to be localised, and so consumption of probiotics to ensure gut microbe balance was to have an impact on digestive health. While this is certainly true, there’s an increasing appreciation of the wide effects. Not only on physical health, but on mental health too – more information in this blog article I wrote towards the end of 2020.
So with such a potential wide impact from disruption to one’s gut microbiota, how can we help our ‘friendly’ bacteria to help us? Keeping personal antibiotic use to an absolute minimum is one way by simply saying no to antibiotics and exploring alternatives (Figure 2). I for one, have not had to treat an infection of mine using antibiotics since 1990. The comprehensive review paper (Ferrer et al., 2017) on the topic of how the functioning of the gut microbiome is negatively impacted by antibiotic therapy reviews several studies that have demonstrated just how bad antibiotics are for our gut microbiota. These authors cover numerous references to studies demonstrating the general reduction in total gut bacteria and also for specific taxa. The complex diagram presented in Figure 3 shows how particular gut bacteria and negatively effected by antibiotics. This really does highlight the magnitude of the (growing) problem around adverse effects on personal health of antibiotic therapy.

Despite this negativity, there are some interesting opposing results. Martín-Núñez et al. (2019) found that the significant changes to the gut microbiota in human subjects with antibiotic therapy for Helicobacter pylori infection improved host glucose homeostatis. So it’s not all doom and gloom, but that doesn’t mean we should be encouraging an imbalance of one’s gut microbiota! Furthermore, Raymond et al. (2016) raises an interesting finding that although negative impacts to the gut microbiome composition take place, they are predictable and with knowledge of an individual’s original gut microbiome profile.
Despite the above consequences in the human context, the fact remains that agricultural use of antibiotics accounts for the vast majority of antibiotic sales, in the US (Figure 4). With the rapidly developing interest in plant-based foods, this will hopefully become less of a problem, but certainly, the absolute dominance of antibiotic use in the context of veterinary medicine as compared with human medicine, is yet another reason to consider plant-based dietary options.

So where does that leave us? Antibiotics have a place, and if they are used for their intended purpose then it is unlikely we would be in the predicament we find ourselves in now. Therefore, we all need to contribute and play our part to halt the deleterious effects on our personal health and on our public health by refusing to be part of antibiotic overuse and misuse.
References
Ferrer, Manuel, Méndez-García, Celia, Rojo, David, Barbas, Coral, & Moya, Andrés. (2017). Antibiotic use and microbiome function. Biochemical Pharmacology, 134, 114–126. https://doi.org/10.1016/j.bcp.2016.09.007
Iizumi, Tadasu, Battaglia, Thomas, Ruiz, Victoria, & Perez Perez, Guillermo I. (2017). Gut Microbiome and Antibiotics. Archives of Medical Research, 48(8), 727–734. https://doi.org/10.1016/j.arcmed.2017.11.004
Martín-Núñez, Gracia Mª, Cornejo-Pareja, Isabel, Coin-Aragüez, Leticia, Roca-Rodríguez, Mª Del Mar, Muñoz-Garach, Araceli, Clemente-Postigo, Mercedes, Cardona, Fernando, Moreno-Indias, Isabel, & Tinahones, Francisco J. (2019). H. pylori eradication with antibiotic treatment causes changes in glucose homeostasis related to modifications in the gut microbiota. PloS One, 14(3), e0213548–e0213548. https://doi.org/10.1371/journal.pone.0213548
Raymond, F., Ouameur, A. A., Déraspe, M., Iqbal, N., Gingras, H., Dridi, B., . . . Corbeil, J. (2016). The initial state of the human gut microbiome determines its reshaping by antibiotics. The ISME Journal, 10(3), 707-720. doi:http://dx.doi.org/10.1038/ismej.2015.148